|
2/18/2023 0 Comments Precipitate rules 
A few of the most popular types of these games include classic slots, progressive slots, and video slots. If you want to try your luck at online slot games, there are several different kinds of these games. These include: (1) convenience of playing the game on mobile phones (2) attractive graphics and sounds and (3) high payout percentage. There are several reasons to play slot online. If you win, you will receive a check in the mail if your prize doesn’t exceed $600, and a form that provides instructions on how to claim your prize. Once subscribed, you can pick the numbers that you want to play, and the system will check the tickets to see if your numbers are among those drawn. Some sites allow you to subscribe for a week, month or even year-long periods. They allow players five chances to win in a single game.Īnother way to play the lottery is to subscribe to a service that will automatically purchase tickets for you. Pull tabs are another type of lottery game that offer quick instant wins. Some online bingo games even feature features that help players find patterns. Bingo, an American lottery-style raffle, is another common online game. In this game, players choose their own lucky numbers without waiting for draws. Keno, a Chinese lottery game, is another example of this. Online scratch-off tickets, for example, allow players to scratch off numbers to win instantly. While some state lotteries are very different from online versions, some of them have features that allow players to play online. 
The grand prizes for both of these games can exceed $1 billion. The two largest nationwide lotteries are MegaMillions and Powerball. Many of these lotteries offer drawing and instant win games. and the US Virgin Islands also run their own lotteries. 1.There are approximately 45 different state lotteries. Here are 5 examples of double replacement reactions. However, since the carbonic acid is unstable it will break down into water and carbon dioxide gas. For example, nitric acid can be reacted with aqueous sodium bicarbonate with the products as shown: In these types of double replacement reactions, one of the products is a gas. This confirms that it is a neutralization reaction. For example:Īfter swapping the ions, it yields water and a compound (NaCl, a salt) as the products. This means that you can recognize a neutralization reaction if after swapping their partners of anions or cations, it yields a compound (usually a solid) and water. It results in two products, water, and salt. Neutralization reactions are reactions between 2 ionic compounds that consist of an acid and a base. However, if one of the compounds is insoluble, then we can predict that it will form the precipitate and the reaction will occur. If both the compounds will dissolve, then the reaction is not possible. This is a chart or table that gives the solubility rules to know which ionic compounds will dissolve and which won’t. We can find out by using the solubility rules. How can we know in advance that a double replacement reaction will occur between two aqueous ionic compounds? One of the products (AX or BY) is an insoluble solid while the other is an aqueous compound.įor example, if you react lead (II) chloride (PbCl₂) solution with a solution of Potassium Sulphate (K₂SO₄), it gives a precipitate and an aqueous compound in the following way. If these react so that AB and XY swap cations, they produce AX and BY. These have the aqueous ions A+, B- (for AB) and X+, Y- (for XY). In these reactions, two aqueous ionic compounds react to produce an insoluble solid product called a precipitate along with another aqueous compound.įor example, we can have AB and XY as the reactants. 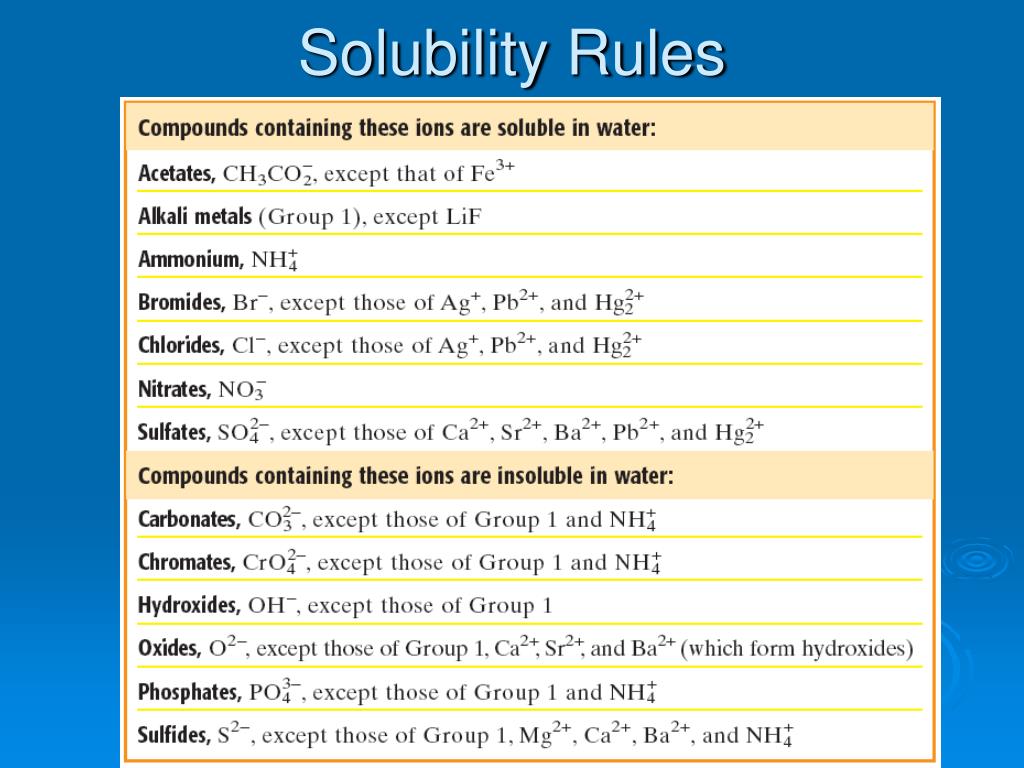
Gas-forming reactions Precipitation reaction.The reaction involves an exchange of cations or anions between the reactants.ĭouble replacement reactions fall into 3 general groups: Two of the compounds are reactants and 2 are products. In a double replacement reaction, there are usually 4 compounds in all. AB becomes AD by giving up B to take up D from CD. 
The reactants AB and CD each give up their partners to the other. This is a reaction between two ionic compounds in which the two swap partners in this way: In a double replacement reaction, the reaction proceeds according to this general form: We also classify the reactions that these substances take part in.ĭouble replacement reactions are just one type of reaction you can get with them. In the chemistry world, we classify many things such as elements and compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
